-
PDF
- Split View
-
Views
-
Cite
Cite
Tianwen Huang, Yueguang Liu, Menggui Huang, Xiaolin Zhao, Leping Cheng, Wnt1-cre-mediated Conditional Loss of Dicer Results in Malformation of the Midbrain and Cerebellum and Failure of Neural Crest and Dopaminergic Differentiation in Mice, Journal of Molecular Cell Biology, Volume 2, Issue 3, June 2010, Pages 152–163, https://doi.org/10.1093/jmcb/mjq008
Close - Share Icon Share
Abstract
The involvement of microRNAs (miRNAs) in the development of the neural crest (NC) cells and other neuronal differentiation is still poorly understood. Here, we investigated the global function of miRNAs in embryonic development by examining the Wnt1-cre-mediated Dicer knockout mice. Dicer ablation resulted in malformation of the midbrain and cerebellum and failure of NC and dopaminergic differentiation. First, the Dicer mutant fetuses exhibited dramatic malformation of the tectum and cerebellum and the eyelids were open. Second, the skeletal structures that are derived from the cranial NC were lost or mostly ablated in Dicer mutant mice. Third, deletion of Dicer in the NC cells resulted in the malformation of the dorsal root ganglia, enteric nervous system and sympathetic ganglia. Interestingly, the expression of neuropeptide Y and its potential regulators TrkA, AP-2α and AP-2β was largely abolished in sympathetic neurons of Dicer mutant mice. Fourth, in situ hybridization data revealed that the expression of miR-9, miR-124 and miR-218 in the midbrain and rostral hindbrain area was mostly eliminated in the Dicer mutant mice. We then demonstrated that the development of dopaminergic neurons was impaired in Dicer-deleted mice. Our studies therefore suggest that miRNAs contribute to the embryonic development in multiple locations.
Introduction
Neural crest (NC) cells are multipotent progenitors that contribute to a variety of peripheral neural tissues and to the development of the head skeleton. NC cells emigrate from the dorsal neural tube during embryonic stages and migrate along stereotypic pathways to defined sites for differentiation (Le Douarin and Kalcheim, 1999). As a population, the NC is of widespread interest due to its plasticity, motility and contribution to developmental disorders and cancers (Knecht and Bronner-Fraser, 2002; Le Douarin et al., 2007; Adams et al., 2008). Extrinsic signaling molecules and intrinsic transcription factors are required for the initial specification and subsequent development of NC cells (Sauka-Spengler and Bronner-Fraser, 2008). MicroRNAs (miRNAs) recently were shown to play important roles in embryonic neural development (Coolen and Bally-Cuif, 2009; Fineberg et al., 2009). However, the involvement of miRNAs in the development of the NC cells is still poorly understood.
miRNAs are a class of small RNAs that bind to specific mRNA targets, leading to mRNA degradation or translational inhibition (Bartel, 2004; Jackson and Standart, 2007). Dicer, an RNase III endonuclease, is the critical enzyme for miRNA biosynthesis. Only one Dicer (Dicer1-Mouse Genome Informatics) gene exists in the mouse genome, which presumably mediates the processing of all miRNAs. In order to reveal general miRNA function, Dicer gene knockout mice were generated. The early lethality of the Dicer null mutation requires the generation of tissue-specific Dicer knockout mice (Bernstein et al., 2003; Cobb et al., 2005; Harfe et al., 2005; Andl et al., 2006; Yi et al., 2006). Since ∼70% of known miRNAs are expressed in the mammalian brain (Cao et al., 2006), it is not surprising that Dicer deletion in the developing mammalian nervous system results in the abnormal development of olfactory neurons, the cortex, the hippocampus, the inner ear, oligodendrocytes and the retina (Choi et al., 2008; Davis et al., 2008; De Pietri Tonelli et al., 2008; Friedman et al., 2009; Kawase-Koga et al., 2009; Soukup et al., 2009; Dugas et al., 2010; Georgi and Reh, 2010; Zhao et al., 2010).
Wnt1 is expressed in the midbrain–hindbrain isthmus region and is critical for the proper formation of the mid- and hindbrain structures (Wilkinson et al., 1987; McMahon and Bradley, 1990; Thomas and Capecchi, 1990). Additionally, Wnt1 is expressed in the NC precursor cells and a Wnt1-cre line has been used efficiently in generating conditional knockouts (CKOs) in the NC cells (Brault et al., 2001; Chen et al., 2006; Taylor et al., 2007). In this study, we examined the function of Dicer in the development of the mid- and hindbrain regions and in NC and dopaminergic neuronal differentiation by analyzing the Wnt1-cre-mediated Dicer knockout mice.
Results
Inactivation of Dicer function by using Wnt1-cre
To study the role of miRNAs in the development of the NC-derived structures, we ablated Dicer by crossing the Dicerflox mice with a well-characterized Wnt1-cre line in which Cre recombinase is expressed in migrating NC cells (Danielian et al., 1998; Yi et al., 2006). To confirm the spatial specificity of Wnt1-cre-mediated recombination, we crossed the Wnt1-cre mice with a reporter line Tau-nLacZ to detect lacZ expression. At E12.5 and E18.5 in developing embryos, lacZ was expressed in the mid-hindbrain, spinal cord, dorsal root ganglia (DRG) and head skeleton (Supplemental Figure S1).
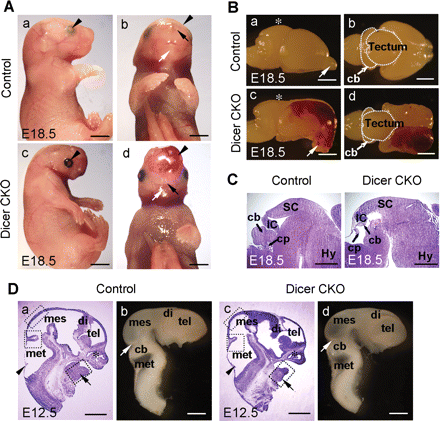
To investigate the consequences of Dicer ablation driven by Wnt1-cre during mouse development, we first examined E18.5 CKO mice. The Dicer mutant fetuses exhibited dramatic brain malformation and the eyelids were open (Figure 1A, a and c). Interestingly, the lower jaw in Dicer knockout mice was absent and there was an abnormal accumulation of blood within the cranial region, though the nostrils and whisker pads were present (Figure 1A, b and d). Macroscopic examination revealed that the olfactory bulbs turned downward and the size of the tectum and the cerebellum was clearly reduced in the Dicer-ablated embryos compared with control littermate brains (Figure 1B). The malformation of the tectum and the cerebellum was corroborated by examination of sagittal brain sections. In the Dicer knockout mice, the cerebellum was almost ablated and the sizes of the rostral tectum (superior colliculus) and caudal tectum (inferior colliculus) were markedly reduced (Figure 1C).
Wnt1-cre-mediated conditional loss of Dicer results in malformation of the midbrain, cerebellum and mandible. (A) Newborn Dicer CKO mice were found with dramatic malformations in the brain region. Compared with the control mice (a and b), the Dicer mutant embryos (c and d) displayed severe craniofacial malformations at E18.5 accompanied by open eyelids (arrowheads in a and c). The most dramatic overall change in the Dicer null mice was the lack of mandible (white arrows in b and d). Ectopic accumulation of blood was also notable in the Dicer mutant (arrowheads in b and d). On the other hand, the nostrils were formed in the Dicer null mice (black arrows in b and d). (B) Lateral (a and c) and dorsal (b and d) views of the dissected E18.5 mouse brain. The tectum of the mutant was underdeveloped (asterisks in a and c) and the olfactory bulbs turned downward (arrows in a and c). The midbrain and cerebellum were smaller in the Dicer mutant (the dashed regions in b and d) and the hematose cortex was clearly visible (c and d). (C) Sagittal HE stained cryosections of the E18.5 brains shown in (B). Note that the cerebellum was mostly ablated in the Dicer mutant, but the choroids plexus was formed. (D). Craniofacial defect was observed at E12.5: (a and c) the HE staining of the sagittal cryosections and (b and d) the lateral view of dissected brains. The mandible process in the mutant was underdeveloped (arrows in a and c), whereas the nasal cavities were formed (asterisks in a and c). The developmental progress of the CNS was similar (compare a, c and b, d); the choroids plexus (arrowheads in a and c) and the isthmus (white arrows in b and d) were positioned correctly. The dashed boxes in panels a and c represent the regions where cell counting experiments were performed in Figure 6. cb, cerebellum; cp, choroids plexus; di, diencephalon; Hy, hypothalamus; IC, inferior colliculus; mes, mesencephalon; met, metencephalon; SC, superior colliculus; tel, telencephalon. Scale bars: 2 mm in A and B; 1 mm in C and D.
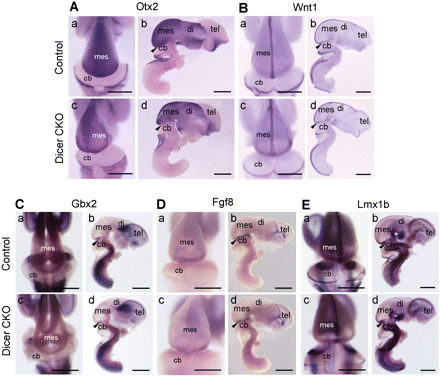
To define the earliest stage at which histological changes were detectable in the Dicer null embryos, HE staining was performed on E12.5 embryos. The major brain regions were formed in the E12.5 Dicer null mice (Figure 1D). This was in line with the analysis of early mid- and hindbrain patterning markers in the Dicer mutant embryos at E12.5 by whole-mount in situ hybridization (Figure 2). However, the reduced size of the lower jaw region was apparent in the Dicer knockout embryos at this stage (Figure 1D, a and c).
Patterning of the mid-hindbrain was normal in the Dicer-ablated mice. Whole-mount in situ hybridization was performed on dissected brains from E12.5 control and Dicer mutant embryos with probes for Otx2 (A), Wnt1 (B), Gbx2 (C), Fgf8 (D) and Lmx1b (E). Dorsal (a and c in A–E) and lateral (b and d in A–E) views are indicated. In each lateral view, one side of the telencephalic vesicle was removed. All genes showed almost identical expression patterns in the control and Dicer null embryos, indicating that the rostral–caudal pattern was unaffected and the MHB organizer was correctly positioned in Dicer mutants. Arrowheads mark the position of the isthmus. cb, cerebellum; di, diencephalon; mes, mesencephalon; tel, telencephalon. Scale bars: 1 mm.
Lack of skeletal structures derived from the cranial NC
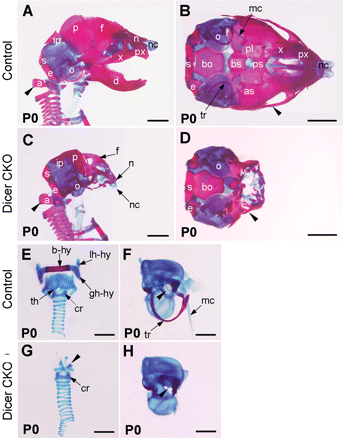
To analyze the bone formation in the head, skeletal preparations from P0 wild-type and Dicer mutant embryos were stained with Alizarin Red and Alcian Blue to reveal the bone and cartilage, respectively. The trunk skeleton, including the vertebral column up to the atlas bone (arrowheads in Figure 3A and C), was unaffected in the Dicer mutant embryos (data not shown). In the head region, fate mapping studies show that the NC gives rise to the skull bones comprising the facial skeleton (e.g. nasal and maxillary), the mandibular skeleton, palatine, squamosal, frontal, nasal, alisphenoid and interparietal (medial portion) (Chai et al., 2000; Jiang et al., 2002; Gross and Hanken, 2008; McBratney-Owen et al., 2008; Yoshida et al., 2008). In addition, the pharyngeal skeleton, including the hyoid bone complex and thyroid cartilage, are also derived from the NC cells (Schoenwolf et al., 2008; Minoux et al., 2009). On the other hand, the otic capsules and several other bones [i.e. parietal, interparietal (lateral portion), supraoccipital, exoccipital and basioccipital] are assigned mesodermal originals (Jiang et al., 2002; McBratney-Owen et al., 2008). Interestingly, skeletal elements of NC origin were absent or severely ablated, whereas the bones from the mesodermal origin were present in the Dicer mutant mice (Figure 3A–H). Other NC derivatives, including the tympanic ring (tr), Meckel's cartilage (mc) and the stapes, were also absent in the Dicer knockout mice (Figure 3F and H).
Craniofacial skeletal defects in Dicer-deficient mice. Skeletons of P0 wild-type (A, B, E and F) and Dicer mutant (C, D, G and H) fetuses were stained with Alizarin Red and Alcian Blue. (A–D) Lateral (A and C) and basal (B and D) views are indicated. The clavicle of both fetuses (A and C) and the mandible bone of the wild type (B) were removed to enhance the view of the laryngeal skeleton (A and C) and cranial base (B). In the mutant, the maxilla (x), premaxilla (px), mandible (d), presphenoid (ps), palatine (pl) bones were lost. The frontal (f) and nasal (n) bone, nasal capsule (nc), basisphenoid (bs), alisphenoid (as) and jugal bones (arrowheads in B and D) were underdeveloped in the mutant. In contrast, the basioccipital (bo), exoccipital (e), supraoccipital (s), lateral portion of the interparietal (ip), parietal (p) and the otic capsule (o) were formed in the mutant. Dissection of the laryngeal skeleton of wild-type (E) and Dicer mutant (G) fetuses revealed that elements of the hyoid bone (hy) were completely lost in the mutant, and the only remaining NCC-derived laryngeal skeleton element was vestigial thyroid cartilage (arrowhead in G). The otic vesicle was present in the Dicer mutant but not well formed (F and H). The stapes was properly positioned into the oval window in wild type but was lost in the mutant (arrowheads in F and H). The tr and mc (arrows in F) were also ablated in Dicer knockout mice. a, atlas bone; b-hy, body of the hyoid bone; cr, cricoid cartilage; gh-hy, greater horn of the hyoid bone; lh-hy, lesser horn of hyoid bone; th, thyroid cartilage. Scale bars: 2 mm in A–D; 1 mm in E–H.
Impaired development of the DRG, enteric nervous system and sympathetic ganglia in Dicer mutant mice
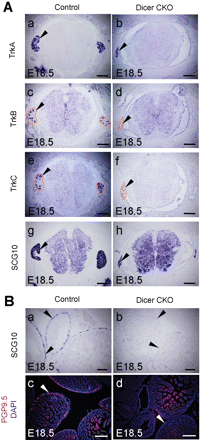
NC cells are multipotent progenitors that contribute to diverse derivatives in the peripheral and enteric nervous system (ENS). Therefore, it was of interest to analyze the development of DRG, the ENS and sympathetic ganglia in the Wnt1-cre; Dicerflox/flox embryos. TrkA, TrkB and TrkC, all of which marks a subpopulation of DRG, and the pan-neuronal molecular marker SCG10 were used to detect the development of DRG. Though the development of DRG in Wnt1-cre; Dicerflox/flox remained largely normal at E14.5 compared with that of control littermates, reduced size of DRG was observed in the Dicer mutants at E18.5 (Figure 4A and data not shown). SCG10 was also expressed in the ENS. However, the expression of SCG10 in the ENS was largely abolished at E18.5 and reduced expression of SCG10 was observed in the ENS at E14.5 (Figure 4B, a and b and data not shown). This was also the case for another ENS molecular marker, PGP9.5 (Figure 4B, c and d and data not shown).
Development of the sensory and ENS in Dicer mutant mice. (A) in situ hybridization was performed on sections of thoracic DRG from control and Dicer mutant at E18.5 with the indicated probes. Dashed lines indicate the contours of the DRG (c–f). Note the reduced size of the DRG in the Dicer mutant embryos (arrowheads). (B) in situ hybridization or immunostaining was performed on sections of enteric neurons from control and Dicer-ablated embryos at E18.5. Note the loss or great reduction of the expression of SCG10 and PGP9.5 in the enteric neurons from Dicer null mice (arrowheads in a, b, c and d). DAPI staining was used to indicate the nucleus of the cells (c and d). Scale bars: 200 µm.
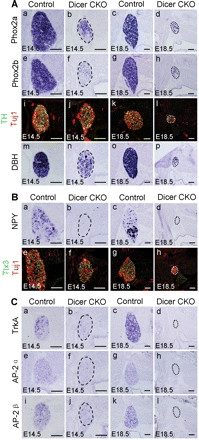
Sympathetic ganglia neurons also originate from the NC. Transcription factors Phox2a and Phox2b are expressed in the sympatho-adrenal precursors and are required for their differentiation. The enzymes tyrosine hydroxylase (TH) and dopamine-β-hydroxylase (DBH) are required for catecholamine synthesis and are characteristic of noradrenergic neurons. The expression of Phox2a, Phox2b, TH and DBH in the sympathetic ganglia of Dicer-ablated embryos was reduced at E14.5, and the size of the sympathetic ganglia was dramatically reduced at E18.5 (Figure 5A). The proportion of TH-positive neurons in the sympathetic ganglia was 88.7 ± 3.3% and 49.2 ± 4.3% (P < 0.001) in the control and Dicer mutant embryos at E14.5, respectively. In order to test whether the Dicer mutation played a role in specifying a subset of sympathetic ganglia neurons, we examined the expression of neuropeptide Y (NPY) and Tlx3 in the Dicer null embryos. NPY and Tlx3 were expressed in the sympathetic ganglia neurons of control mice at E14.5. However, in the E14.5 Dicer mutant embryos, the expression of NPY in the sympathetic ganglia neurons was almost eliminated, whereas the expression of Tlx3 remained (Figure 5B, a, b, e and f). The proportion of neurons that expressed Tlx3 in the Dicer mutant mice (69.2 ± 7.2%) was even higher (P < 0.01) than in the wild-type mice (44.3 ± 5.3%). The expression of NPY was mostly eliminated in the sympathetic ganglia neurons of Dicer mutants at E18.5 compared with the control mice, and the expression of Tlx3 was still present (Figure 5B, c, d, g and h). Because the transcriptional activity of the NPY gene was subject to the regulation of nerve growth factor (NGF) and transcription factor AP-2α (Li et al., 2000), we examined the expression of the NGF receptor TrkA and transcription factor AP-2α and its homolog AP-2β (Hong et al., 2008) in the Dicer mutant mice. The expression of TrkA, AP-2α and AP-2β was largely abolished in both the E14.5 and the E18.5 Dicer null embryos (Figure 5C), which may underlie the selective elimination of NPY in Dicer mutant mice.
Impaired development of the sympathetic ganglia in Dicer-ablated mice. In situ hybridization or immunostaining was performed on sections of stellate ganglia from control and Dicer-ablated embryos at E14.5 or E18.5 with the indicated probes or antibodies. The expression of Phox2a (A, a–d), Phox2b (A, e–h), TH (green)/Tuj1 (red, A, i–l) and DBH (A, m–p) was reduced in E14.5 Dicer mutants and greatly reduced at E18.5 in Dicer null mice. NPY and Tlx3 were expressed in subpopulations of sympathetic neurons (B, a–h). In the Dicer mutants, the expression of NPY was mostly ablated (B, a–d) though the expression of Tlx3 was proportionally remained (B, e–h). The expression of Tlx3 is shown in green and the expression of Tuj1 is shown in red. Consistent with the NPY expression pattern, the expression of its potential transcriptional regulators, TrkA (C, a–d), AP-2α (C, e–h) and AP-2β (C, i–l) was almost ablated in the Dicer knockout mice at both E14.5 and E18.5. Scale bars: 100 µm.
The development of DRG and sympathetic ganglia are closely related to NC migration. The NCs migrate to a position between the somite and neural tube and begin their condensation into DRG ganglion primordia around E9–E9.25, which is reflected by the expression of neurogenin 1 (Ma et al., 1999). The NCs arrive along the dorsal aorta to form the primary sympathetic ganglion chain by E10–E10.5. These cells subsequently undertake a second migratory step from the dorsal aorta to their final destination (Hirsch et al., 1998; Huber, 2006). The normal size of DRG and the sympathetic ganglia in the Dicer knockout mice at E12.5 indicated that the NC cell migration was largely normal in the Dicer mutant mice (data not shown).
Increased apoptosis in Dicer mutant mice
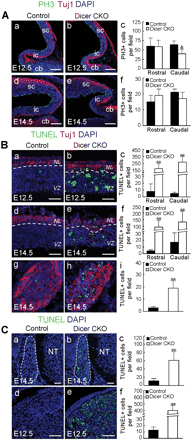
The abnormal development in the Dicer mutant mice prompted us to examine whether the proliferation of the progenitors was impaired or if apoptosis played a critical role. Quantification of the mitotic figures identified by immunostaining for phosphohistone H3 showed that the abundance of mitotic progenitors was largely the same in the mid- and hindbrain regions from control and Dicer-ablated mice at E12.5 and E14.5 (Figure 6A, a–f). This was supported by the cumulative BrdU-labeling experiments in which the numbers of BrdU-positive cells in the mid- and hindbrain of control and Dicer-ablated embryos at E12.5 were similar (data not shown). We also did not observed the difference in the rate of proliferation in DRG and sympathetic ganglia from control and Wnt1-cre-mediated Dicer-knockout embryos at E12.5 (P > 0.05) and E14.5 (data not shown): the percentage of PH3+ cells in DRG at E12.5 from control and Dicer mutant mice was 7.4 ± 2.3% and 6.3 ± 2.2%, respectively and the percentage of PH3+ cells in sympathetic ganglia at E12.5 from control and Dicer mutant embryos was 18.4 ± 5.8% and 14.7 ± 3.6%, respectively. Instead, TUNEL staining showed that Dicer ablation resulted in apoptosis, which was observed in the tectum, sympathetic ganglia, DRG and mandible area of the Dicer mutant embryos (Figure 6B and C). These lines of data agree with a recent study showing that Dicer is required for the survival of the differentiating NC cells (Zehir et al., 2010). The massive apoptosis in the Dicer-ablated ventricular zone and the neuronal layer suggested that the apoptosis took place in both the mitotic progenitor cells and the post-mitotic neurons (Figure 6B, a–f).
Apoptosis occurred in the mid-hindbrain and NC-derived cells in Dicer mutant mice. (A) Immunostaining for phosphohistone H3 (PH3, green), Tuj1 (red) and DAPI (blue) was performed on sagittal sections through the rostral midbrain (Rostral) and caudal midbrain and cerebellum (Caudal) from the control and Dicer mutant embryos at E12.5 (a and b) and E14.5 (d and e). The quantification of mitotic (PH3-positive) apical progenitors at E12.5 (c) and E14.5 (f) is indicated. △: P = 0.056. cb, cerebellum; ic, inferior colliculus; sc, superior colliculus. (B) TUNEL (green), Tuj1 (red) and DAPI (blue) staining was performed on sagittal sections of tectum (a, b, d and e) or transverse sections of thoracic sympathetic ganglia (g and h) from control and Dicer mutant embryos. Note the dramatically increased apoptosis in the Dicer-ablated embryos (c, f and i). (C) TUNEL (green) and DAPI (blue) staining was performed on transverse sections of the lumbar DRG at E14.5 (a and b) or sagittal sections of the mandible process (d and e) from control and Dicer mutant embryos at E12.5. Dashed lines in a and b indicate the contour of DRG. Note that apoptosis occurred in the Dicer-ablated embryos (c and f). **P < 0.001. NL, neuronal layers; NT, neural tube; VZ, ventricular zone. Scale bars: 150 µm in A; 50 µm in B and C.
Dicer is required for the specification of dopaminergic neurons
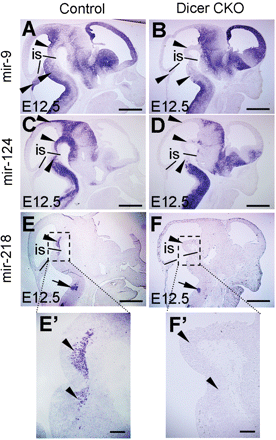
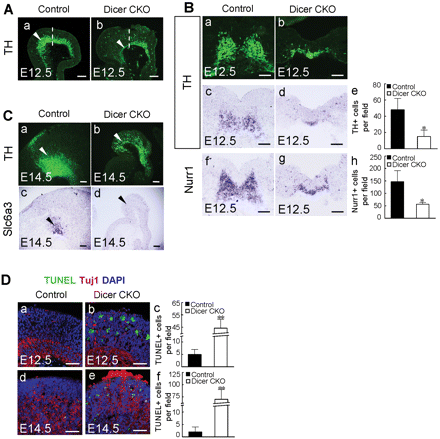
To ascertain the Wnt1-cre-mediated ablation of Dicer activity, we investigated whether the expression of mature miRNAs was affected in the Dicer CKO embryos. For this purpose, we performed in situ hybridization using locked-nucleic-acid modified (LNA) probes, an approach that has been shown to reveal the presence of mature miRNAs (Kloosterman et al., 2006). MiR-9, miR-124 and miR-218 were expressed in the E12.5 wild-type mouse brain. Interestingly, the expression of these miRNAs was largely eliminated in the tectum, ventral midbrain and rostral hindbrain regions of Wnt1-cre-mediated Dicer mutant mice at E12.5 (Figure 7A–F). Loss of the expression of the miRNAs in the Dicer knockout mice demonstrated that deletion of Dicer was functional. On the other hand, abrogation of the expression of the miRNAs in the ventral midbrain prompted us to examine the consequences of the Dicer and miRNA ablations in this brain region. Because some dopaminergic neurons develop from the ventral midbrain, we examined the development of midbrain dopaminergic (mDA) neurons in the Dicer null embryos. TH expression in the ventral midbrain was dramatically reduced in the E12.5 Dicer-ablated embryos. This was particularly evident in the caudal region where the expression of TH was largely abolished (Figure 8A, a and b). Cell counting of the TH and Nurr1-positive cells confirmed that the differentiation of mDAs was impaired in Dicer mutant embryos at E12.5 (Figure 8B, a–h). The number of TH-expressing neurons was dramatically reduced in the Dicer mutant mice at E14.5 when compared with the control embryos (Figure 8C, a and b). Remarkably, the TH-positive cells did not express another mDA neuron-specific marker gene, the dopamine transporter Slc6a3, in the Dicer null mice (Figure 8C, c and d). This result indicated that an mDA precursor still develops in the ventral midbrain of the Dicer mutant mice, but this precursor does not properly differentiate into an mDA neuron in the absence of Dicer, as judged by the lack of Slc6a3 expression. The mDA precursors that did not execute their correct differentiating program may go to die, which was in line with the fact that apoptosis occurred in the differentiating ventral midbrain of Dicer-ablated embryos (Figure 8D).
Specific loss of miRNA expression in E12.5 Dicer conditional mutant mice. miRNA in situ hybridization was performed on sagittal sections of control or Dicer-ablated embryos at E12.5 with LNA antisense probes for mir-9 (A and B), mir-124 (C and D) and mir-218 (E and F). (A and B) The expression of mir-9 was specifically ablated in the ventral midbrain and rostral hindbrain of the Dicer mutant (arrowheads). (C and D) The expression of mir-124 was absent in the tectum, ventral midbrain and rostral hindbrain of the Dicer mutant (arrowheads). (E and F) The expression of mir-218 was ablated in the ventral midbrain and rostral hindbrain of the Dicer null mice (arrowheads), whereas the expression of mir-218 in the caudal hindbrain region was unaffected (arrows in E and F). E′ and F′ are higher magnification of the boxed areas in E and F, respectively. The black lines in A–F indicate the mid-hindbrain boundary. is, isthmus. Scale bars: 500 µm in A–F; 100 µm in E′ and F′.
Dicer is required for the differentiation and survival of mDA neurons. (A) Immunostaining with anti-TH antibody was performed on sagittal sections of the ventral midbrain from control and Dicer mutant embryos at E12.5. Note that in the Dicer mutant, the expression of TH was greatly reduced in the caudal portion of the ventral midbrain (arrowheads in a and b). Dash lines indicate the plane of sections shown in B (a and b). (B) Immunostaining with anti-TH antibody (a and b) or in situ hybridization with antisense probes for TH (c and d) or Nurr1 (f and g) was performed on coronal sections of the ventral midbrain from control and Dicer mutants at E12.5. Quantification of TH and Nurr1-positive cells is presented in e and h (*P < 0.05). (C) Immunostaining with anti-TH antibody (a and b) or in situ hybridization with probe for the dopamine transporter Slc6a3 was performed on sagittal sections of the ventral midbrain at E14.5. Note that the expression of TH was detectable (arrowheads in a and b) whereas the expression of Slc6a3 was lost in the Dicer mutant (arrowheads in c and d). (D) TUNEL (green), Tuj1 (red) and DAPI (blue) staining was performed on the sagittal sections of the ventral midbrain from control and Dicer mutant embryos at E12.5 (a and b) and E14.5 (d and e). Quantifications of TUNEL-positive cells are indicated in c and f (**P < 0.001). Scale bars: 100 µm in A–C; 50 µm in D.
Discussion
While the present study was being prepared for publication, a most recent study reported that Dicer is required for the survival of differentiating NC cells (Zehir et al., 2010). In both studies, Dicer was shown to play important roles in the proper development of NC cells. In addition, there are some new findings in our study. First, the cerebellum was almost ablated and the sizes of the tectum were markedly reduced in Wnt1-cre-mediated Dicer knockout mice. Second, the skeletal structures that are derived from the cranial NC (CNC) were clearly demonstrated to be lost or underdeveloped in the Dicer mutant mice. Third, though Dicer was shown to be required for survival of differentiating sympathetic neurons in both studies, we further provided evidence that the expression of NPY, a molecular marker for subsets of sympathetic neurons, and its potential regulators TrkA, AP-2α and AP-2β was specifically abolished in sympathetic neurons of Dicer mutant mice. The data indicated that miRNAs may contribute to the differentiation of sympathetic ganglia in addition to the survival of differentiating sympathetic neurons. Fourth, we found that miR-9, miR-124 and miR-218 were expressed in the E12.5 wild-type mouse brain, and the expression of these miRNAs was largely eliminated in the mid- and hindbrain regions of Wnt1-cre-mediated Dicer mutant mice. We then demonstrated that miRNAs were required for the differentiation and survival of dopaminergic neurons.
miRNAs are involved in the development of the mid- and hindbrain regions
The development of the mid- and hindbrain is controlled by the activity of the isthmic organizer (IsO), which is located at the boundary between the prospective mid- and hindbrain [i.e. the mid-hindbrain boundary (MHB)]. Several transcription and secreted factors, such as Otx2, Gbx2, Fgf8, Wnt1, En1, En2, Pax2 and Lmx1b, constitute a regulatory network controlling the activity of the IsO and the development of the mid- and hindbrain (Liu and Joyner, 2001; Wurst and Bally-Cuif, 2001; Nakamura et al., 2005). miRNA recently emerges as a new player in controlling the development of the IsO and the mid- and hindbrain (Giraldez et al., 2005). In zebrafish, miR-9 directs the late organizer activity of the MHB by targeting the fibroblast growth factor (Fgf) signaling pathway. Moreover, miR-9 promotes neurogenesis near the MHB organizing center (Leucht et al., 2008). In Pax2-cre conditional Dicer knockout mice, notable defects in mid- and hindbrain development that include ablation of the cerebellum and tectum were observed. In this study, we found that the Wnt1-cre conditional Dicer ablation resulted in the malformation of the mid- and hindbrain. Meanwhile, the expression of miR-9, miR-124 and miR-218 was eliminated around the MHB organizing center, indicating that these miRNAs may contribute to the impaired development of the mid- and hindbrain that was observed in the Pax2- and Wnt1-cre-directed Dicer knockout mice.
miRNAs are required for the development of the head skeleton
Much of the skull and the entire pharyngeal skeleton (including the jaws, hyoid and gill structures) have a unique embryonic origin from the CNC, unlike the more posterior axial and appendicular skeletons, which are derived from mesoderm (Helms and Schneider, 2003; Knight and Schilling, 2006). Some paracrine and transcription factors are required for the differentiation of the CNC and development of the head skeleton (Santagati and Rijli, 2003; Chai and Maxson, 2006; Jeong et al., 2008). miRNA recently emerged as a new player in the head skeleton formation. miR-140 was reported to be required for palatogenesis by negatively regulating the platelet-derived growth factor signaling in zebrafish (Eberhart et al., 2008). In this study, we found that all of the skeletal structures that are derived from the CNC were lost or underdeveloped in the Wnt1-cre-mediated Dicer mutant mice. It will be of interest in the future to investigate which individual miRNAs play critical roles in the development of the head skeleton.
miRNAs are required for the development of trunk NC cells
At the trunk level, NC cells contribute to the development of the peripheral and ENS and sympathetic ganglia (Le Douarin and Kalcheim, 1999). In this report, we found that deletion of Dicer in the NC cells resulted in the malformation of the DRG, ENS and the sympathetic ganglia. These data are consistent with a recent report stating that Dicer is required for the survival of differentiating NC cells (Zehir et al., 2010).
Induction of apoptosis in the nervous system is a common feature of most Dicer CKO mice studied thus far (Kim et al., 2007; Schaefer et al., 2007; Davis et al., 2008; De Pietri Tonelli et al., 2008; Coolen and Bally-Cuif, 2009). Indeed, this was also the case with the Wnt1-cre-mediated Dicer knockout mice (Zehir et al., 2010 and this study). On the other hand, in this study we found that Dicer was required for the expression of molecular markers for subsets of sympathetic neurons. In the Wnt1-cre directed Dicer mutant mice, the expression of NPY and its potential regulators TrkA, AP-2α and AP-2β was largely abolished, whereas the expression of another molecular marker Tlx3 for the sympathetic neurons was proportionally retained. It warrants further studies to demonstrate whether miRNAs play important roles in the neuronal specification in the differentiating sympathetic ganglia neurons.
miRNAs are required for dopaminergic neuronal differentiation
The mDA neurons are involved in the control of voluntary movements and in the regulation of emotion-related behavior. They are affected in many neurological and psychiatric disorders, including Parkinson's disease, depression and schizophrenia. Interest in the development of mDA neurons has been heightened by the beneficial effect of DA-neuron transplantation in Parkinson's disease. Several extrinsic signaling pathways and many transcription factors have been shown to be required for proper mDA development (Goridis and Rohrer, 2002; Ang, 2006; Prakash and Wurst, 2006; Smidt and Burbach, 2007). A previous study showed that specific deletion of Dicer in post-mitotic dopaminergic neurons leads to a progressive loss of mDAs (Kim et al., 2007). However, whether miRNAs play important roles in dopaminergic neuronal differentiation remains unknown. In this study, we provided evidence that the differentiation of dopaminergic neurons was impaired in the Wnt1-cre-mediated Dicer mutant mice. Additionally, the absence of the expression of miR-9, miR-124 and miR-218 in the ventral midbrain of Dicer mutant mice warrants studies to reveal whether these miRNAs contribute to the dopaminergic neuronal differentiation.
Materials and methods
Generation of conditional Dicer mutants
Mutant mice carrying a floxed allele of Dicer (Dicerflox/+) were obtained from the Mutant Mouse Regional Resource Centers (MMRRC) by the kind donation of Dr. Alexander Tarakhovsky at the Rockefeller University (Yi et al., 2006). Dicerflox/+ mice were crossed to transgenic mice expressing Cre recombinase under the control of the Wnt1 promoter (Danielian et al., 1998). Double heterozygous progenies (Wnt1-cre; Dicerflox/+) were identified and mated with Dicerflox/flox or Dicerflox/+ mice to obtain Wnt1-cre; Dicerflox/flox CKO embryos, whereas littermates (which carry the incomplete combination of the above alleles) served as wild-type controls. The morning that vaginal plugs were observed was designated E0.5. Wnt1-cre transgenic mice were identified with primers 5′-GCCTGCATTACCGGTCGATGC-3′ and 5′-CAGGGTGTTATAAGCAATCCCC-3′, which were used for detecting the Cre expression. Dicer alleles were genotyped with the primer pair 5′-AAAGCAGAACTCTAATGCCCC-3′ and 5′-AGGTGCCTTTCGTTTAGGAAC-3′ (product sizes: 382 bp from the Dicerflox/+ allele and 244 bp from the wild-type Dicer locus). Tau-nLacZ reporter line was kindly provided by Dr. Silvia Arber (University of Basel, Basel, Switzerland) (Hippenmeyer et al., 2005). All animal procedures are contained in the protocols reviewed and approved by the Animal Care Committees at the Institute of Neuroscience, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences.
In situ hybridization
Section in situ hybridization with digoxigenin-labeled cRNA probes was performed as previously described (Birren et al., 1993). Briefly, following pretreatments (proteinase K, post-fixation and acetic anhydride) sections were prehybridized for 3–4 h at 67°C. Hybridization was carried out for 12–16 h using 1 µg/ml of probe in plastic slide mailers containing sufficient probe solution to immerse the sections. The alkaline phosphatase reaction product was developed using NBT/BCIP reagents, and development was carried out depending upon the abundance of the target mRNA. Whole-mount in situ hybridization followed a protocol by Wilkinson (1992) with some modifications. Briefly, E12.5 embryos were fixed in 4% paraformaldehyde and the dissected brains were pretreated with proteinase K. After re-fixation, the brains were prehybridized at 67°C for 3–4 h and hybridized for 12–16 h. Alkaline phosphatase activity was detected by NBT/BCIP.
The Wnt1 and Fgf8 in situ probes were amplified using gene-specific sets of PCR primers and genomic DNA templates prepared from P21 mouse tail lysate according to previous studies (Parr et al., 1993; Crossley and Martin, 1995). The primers were as follows: Wnt1, 5′-ACGAGTGTCTATGAGGTGCCG-3′ and 5′-TCCATTGGAAGGGAAGAGGAC-3′, 1007 bp; and Fgf8, 5′-CAAAGGCAAGGACTGCGTATTCACA-3′ and 5′-CAGCCAGTGGAATACAAAAATAGAG-3′, 482 bp. Other in situ probes included were described previously (Ma et al., 1999; Qian et al., 2001; Cheng et al., 2003; Gray et al., 2004; Huang et al., 2008).
miRNA in situ hybridization on cryosections was performed using 3′-digoxigenin-labeled LNA antisense probes (Exiqon) to mouse miR-9, miR-124 and miR-218 according to the manufacture's instructions.
Immunostaining
For immunohistochemistry, cryosections were incubated overnight at 4°C with appropriate primary antibody diluted in 0.4% Triton X-100 and 10% heat-inactivated lamb serum in PBS. Sections were then extensively washed in PBS plus 0.1% Tween 20 and incubated for 1 h at room temperature with a secondary antibody conjugated to a fluorochrome. Sections were then washed and mounted in Mowiol (Calbiochem). For DAPI staining, the sections were incubated with DAPI (1 µg/ml, Invitrogen) for 3 min at room temperature. For BrdU-labeling experiments, BrdU (Sigma; 75 µg/g body weight) was injected intraperitoneally at various stages. Embryos were isolated 24 h after injection.
The following antibodies were used: rabbit anti-Tlx3 (1:1000), which was produced against the purified His6-Tlx3 antigen (kindly provided by Drs. Thomas Müller and Carmen Birchmeier, Max-Delbrück-Center for Molecular Medicine, Berlin, Germany) (Muller et al., 2005); rabbit anti-TH (1:500, Pel-Freez); mouse anti-PGP9.5 (1:100, AbD Serotec); mouse anti-TUJ1 (1:1000, Covance); rabbit anti-phosphorylated histone H3(Ser10) (1:500, Millipore); goat anti-mouse Cy3 (1:500, Jackson ImmunoResearch Laboratories) and goat anti-rabbit Alexa-488 (1:300, Invitrogen).
Cell counting
Cryosections of control and Wnt1-Cre; Dicerflox/flox embryos were analyzed by Leica TCS SP2 confocal fluorescence microscopy. PH3-positive cells aligned along the apical region of the ventricular surface were counted. Considering the morphological changes of the caudal midbrain in E14.5 Dicer mutant embryos, the number of PH3+ cells per field at E14.5 (Figure 6A, d and e) was defined as the total PH3+ cells per 400 µm of the apical line. Values were presented as mean ± s.e.m. TUNEL-positive cells from two or three pairs of control and Dicer mutant embryos were counted. TH+, Tlx3+ and Tuj1+ cells in the stellate sympathetic ganglia at E14.5 were calculated from three pairs of control and Dicer mutant embryos. Nine sets of transverse sections from three pairs of control and Dicer mutant mice with positive in situ hybridization signal were used to count neurons that express TH or Nurr1 mRNA in the ventral midbrain. Positive cells with clear nuclei were counted. Values are presented as the mean ± s.d. The differences in values were considered significant at P < 0.05 by Student's t-test.
TUNEL staining on cryostat sections
Apoptotic cells in the developing brain were analyzed by the TUNEL assay using the ApopTag-plus Fluorescein In Situ Apoptosis Detection Kit (Chemicon International). Cell staining was performed according to the manufacturer's protocols.
Skeletal preparation
The skeletal preparation and the Alizarin Red and Alcian Blue staining of skeletons followed standard techniques (Nagy et al., 2003) with minor modifications. Briefly, the newborn mice were eviscerated, fixed overnight in ethanol and then macerated in acetone for 24 h. The fixed fetuses were stained with Alcian Blue 8GX (150 mg/l in 80% ethanol, 20% acetic acid) for 48 h at 37°C, washed in 70% ethanol for 8 h and cleared in 1% KOH overnight. They were then incubated in Alizarin Red S (50 mg/l in 2% KOH) for 6–8 h, cleared in 1% KOH and stored in 50% glycerol.
Funding
This work was supported by grants from the Ministry of Science and Technology of China (2007CB947102 and 2006CB943900), the National Natural Science Foundation of China (30540014, 30770697) and the Science and Technology Commission of Shanghai Municipality (07pj14098).
Acknowledgments
We thank Dr. Andrew P. McMahon at the Harvard University for sharing the Wnt1-cre mice and Dr. Silvia Arber for providing the Tau-nLacZ indicator mouse strain, Dr. Alexander Tarakhovsky at the Rockefeller University and MMRRC for providing the Dicer conditional allele.
Conflict of interest: none declared.
References
Author notes
T.H. and Y.L. contributed equally to this work.